Some Breast Implants May Cause Cancer
Much of the information presented here is from the FDA website on the current recommendations and understanding of breast implant associated cancer.
Individuals with breast implants have a risk of developing breast implant-associated anaplastic large cell lymphoma, or BIA-ALCL. BIA-ALCL is a type of non-Hodgkin’s lymphoma (cancer of the immune system). Like other types of cancer of the immune system, the disease can result in patient deterioration, cancer spread, and patient death. In most cases, BIA-ALCL is found in the scar tissue and fluid near the implant but can be widespread.
Background
In 2011, the FDA identified a possible association between breast implants and the development of anaplastic large cell lymphoma (ALCL).
At this time, most data suggest that BIA-ALCL occurs more frequently following implantation of breast implants with textured surfaces rather than those with smooth surfaces.
- The Australian Therapeutic Goods Administration (TGA) reported a detailed analysis of the 46 patients with confirmed cases of BIA-ALCL in Australia, including the deaths of 3 women. As of September 2016, TGA has confirmed 10 additional cases in Australian patients.
- As of September 30, 2017, the FDA had received a total of 414 medical device reports (MDRs) of BIA-ALCL, including the deaths of nine patients.
272 of the 414 reports included information on the surface information of the implant at the time of the report, including 242 with textured surfaces and 30 with smooth surfaces. 413 of the 414 reports included information on implant fill types. Of these, 234 reported implants were filled with silicone gel and 179 reported implants filled with saline. While the MDR reports provide information regarding the implant at the time of BIA-ALCL diagnosis, they do not typically give information about a patient’s history of breast implants. In the MDR reports, half of the reported cases were diagnosed within 7-8 years of implantation. It is important to note that at the time of diagnosis, patients may have their original breast implants or they may have had one or more replacements.
Medical Literature
Most of the cases reported in the literature describe individuals who have had textured implants. Several recent journal articles explore possible risk factors for developing BIA-ALCL, including the methods used to create the textured surface and the role of biofilm. Most of the published information about treatment describes removal of the implant and the capsule surrounding the implant, and in some patients, treatment with chemotherapy and radiation.
Several recent publications have estimated the risk of developing BIA-ALCL in individuals with textured breast implants. The FDA reports that BIA-ALCL may develop in 1 in between 3,817 to 30,000 women with textured breast implants.
As of this date, the risk appears to be very small and associated with textured breasts implants.
Dr. Kenneth Hughes, Los Angeles plastic surgeon, offers smooth implants of saline, silicone, and gummy bear variety to greatly reduce this risk of cancer and patient deaths.
It is very important to discuss this risk with patients who are undergoing breast augmentation in general, particularly if the patient is undergoing textured breast implant placement.
Medicine is constantly evolving and what may be recommended at one time due to obvious benefit, may need to be altered when and if new information presents.
Dr. Kenneth Hughes of Hughes Plastic Surgery stays abreast of all of the newest information in all aspects of cosmetic plastic surgery, so that patients do not have to review all of the literature to feel informed.
https://www.drkennethbenjaminhughesmd.com
https://www.drkennethhughes.com
To learn more about Dr. Kenneth Hughes, Harvard-trained, board-certified plastic surgeon in Los Angeles, please visit
https://www.hughesplasticsurgery.com/los-angeles-kenneth-hughes/
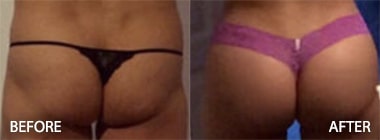
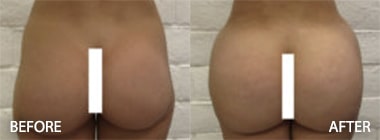
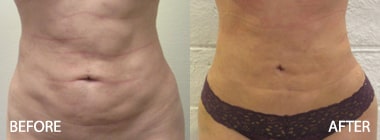
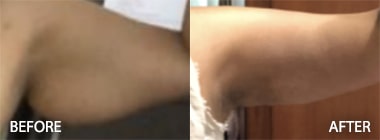
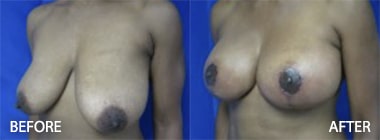
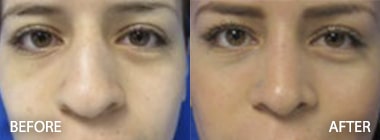
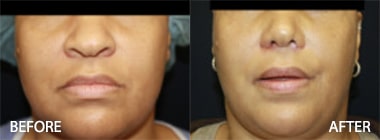
To see before and after photos of Kenneth Hughes, please visit https://www.hughesplasticsurgery.com/los-angeles-before-after-photos/
To contact Kenneth Hughes’s office, please visit
https://www.hughesplasticsurgery.com/contact-los-angeles-plastic-surgeon/